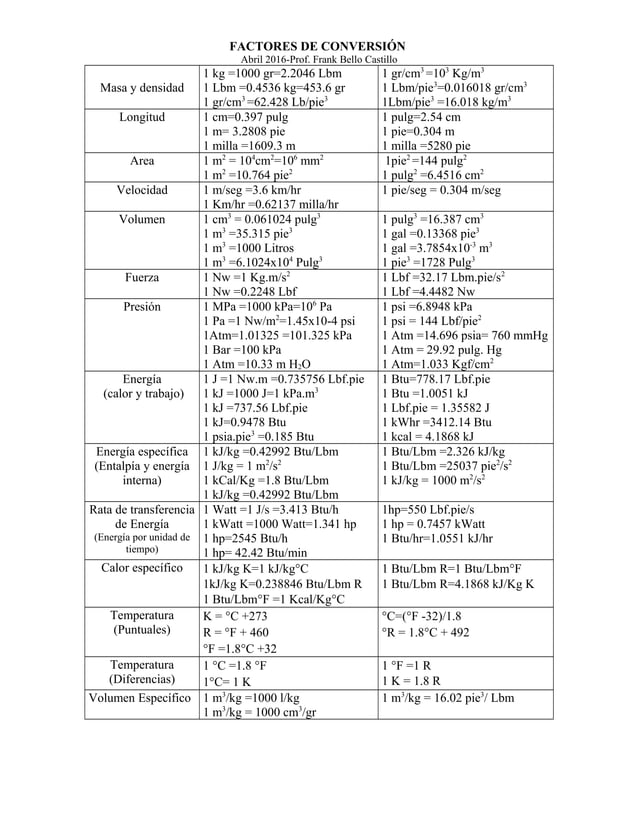
a) Calculate the heat that must be supplied to raise the temperature of 2 kg of water from 25^(@)C to its boiling point at atmospheric pressure.The average specific heat of water in

SOLVED: Queetkn1 In the cylinder of an air motor the compressed air has specific internal energy of 420 kJ/kg at the beginning of the expansion and specific internal energy of 200 kl/kg

An electric heater of power 600 W raises the temperature of 4.0 kg of a liquid from 10.0 ^o C to 15.0 ^o C in 100 s. Calculate the specific heat capacity of liquid: