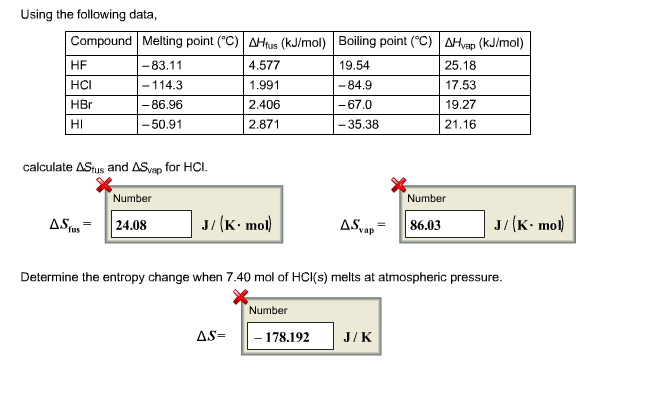
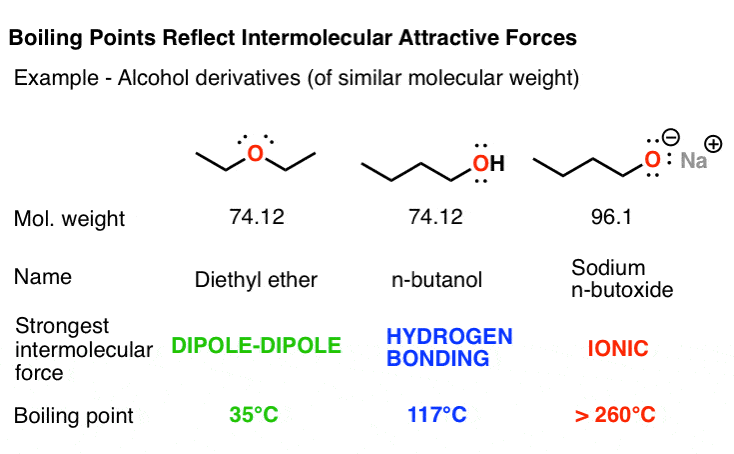
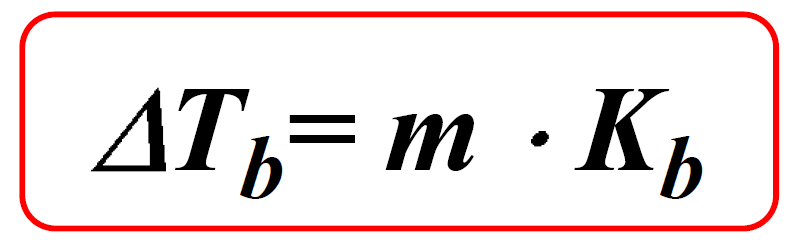Colligative Properties - Boiling Point Elevation, Freezing Point Depression & Osmotic Pressure - YouTube

SOLVED: Calculate the melting point of benzoic acid (C6H5CO2H), given the following data: ΔHfusion = 17.3 kJ/mol and ΔSfusion = 43.8 J/(mol K). Select one: a. 4250 K b. 425 K c. 0.395 K d. 395 K

The melting point of phenol is `40^(@)C`. A solution containting `0.172 g`acetanilide `(C_(8)H_(9)OH - YouTube

SOLVED: Calculate the melting point of ice under a pressure of 1OMPa: Assume that the density of ice under these conditions is approximately 0.915 gcm 3 and that of liquid water is

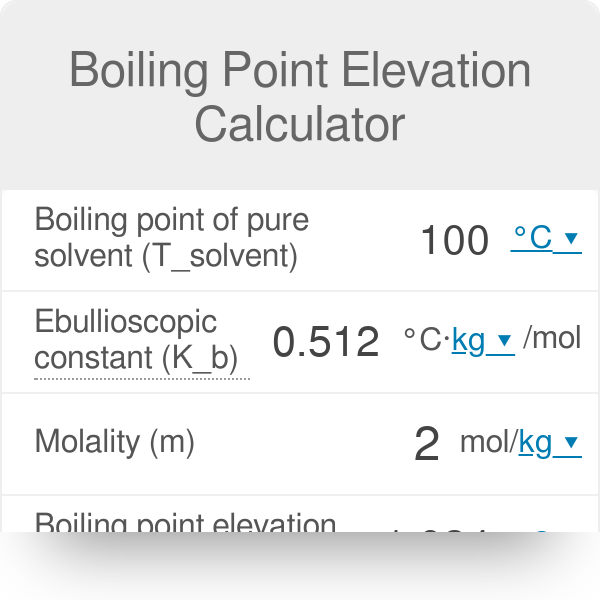
Calculate the freezing point and the boiling point at 1 atmosphere of a solution containing 30 g cane sugar (molecular mass 342 ) and 150 g water.Given : Kb = 0.513 and Kf = 1.86