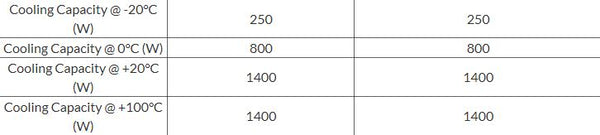
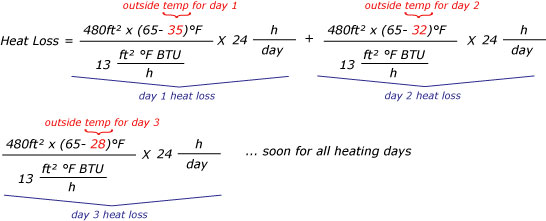
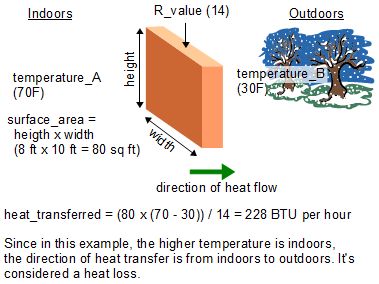
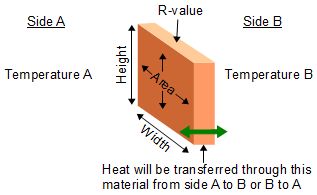
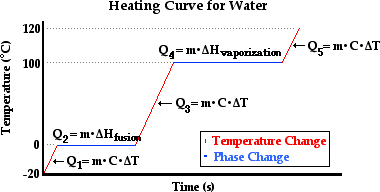
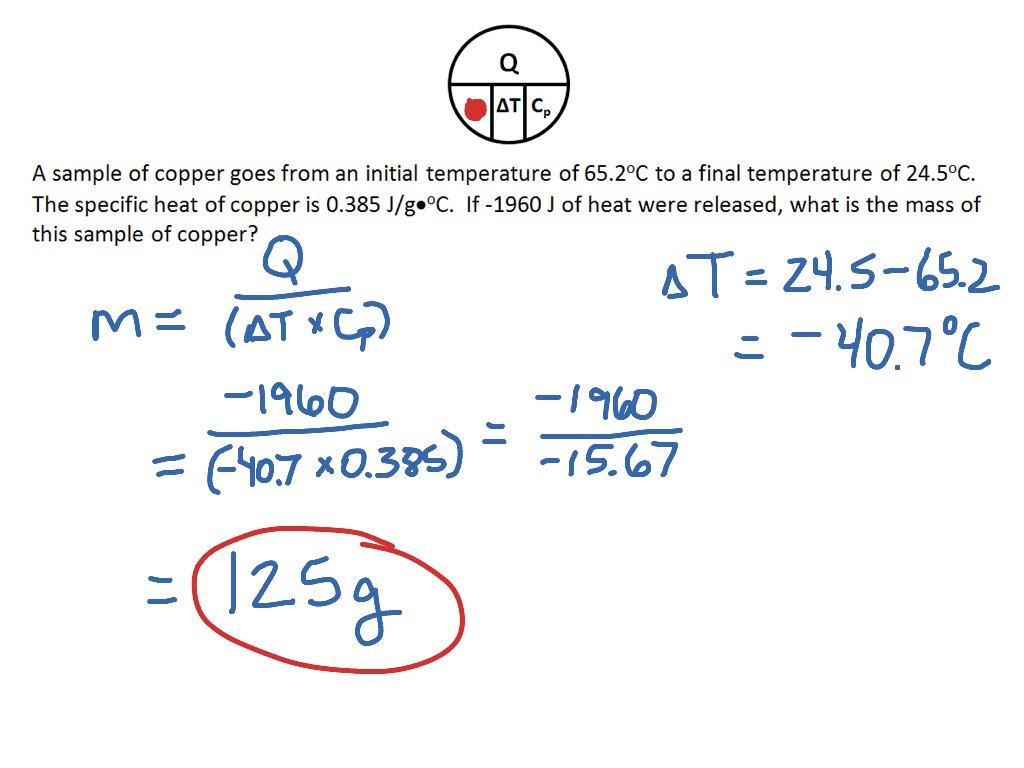
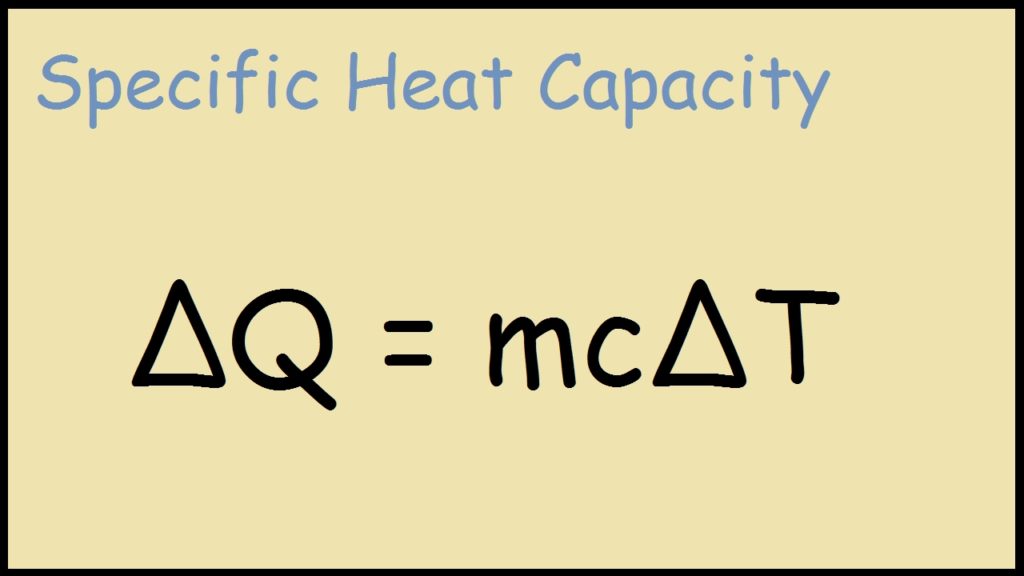How to determine the heat evolved or consumed by the reaction of 1.0 g SO2(g) with excess oxygen, with results from a Hess' Law Equation? | Socratic
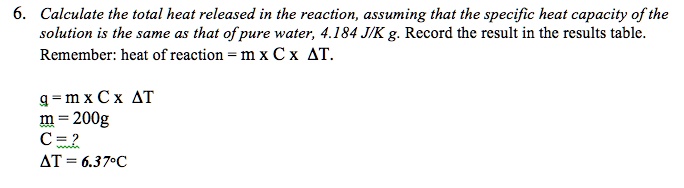
SOLVED: Calculate the total heat released in the reaction, assuming that the specific heat capacity of the solution is the same as that of pure water, 4.184 JIK g. Rccord thc rcsult

Heat Capacity and Specific Heat Calculations - Practice - A 1 g sample of decane, C 10 H 22 , is - Studocu