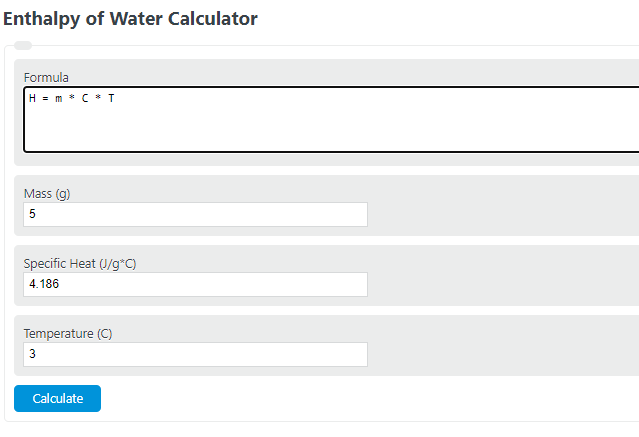
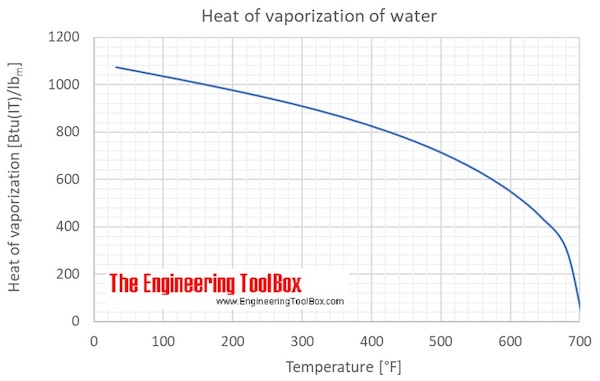
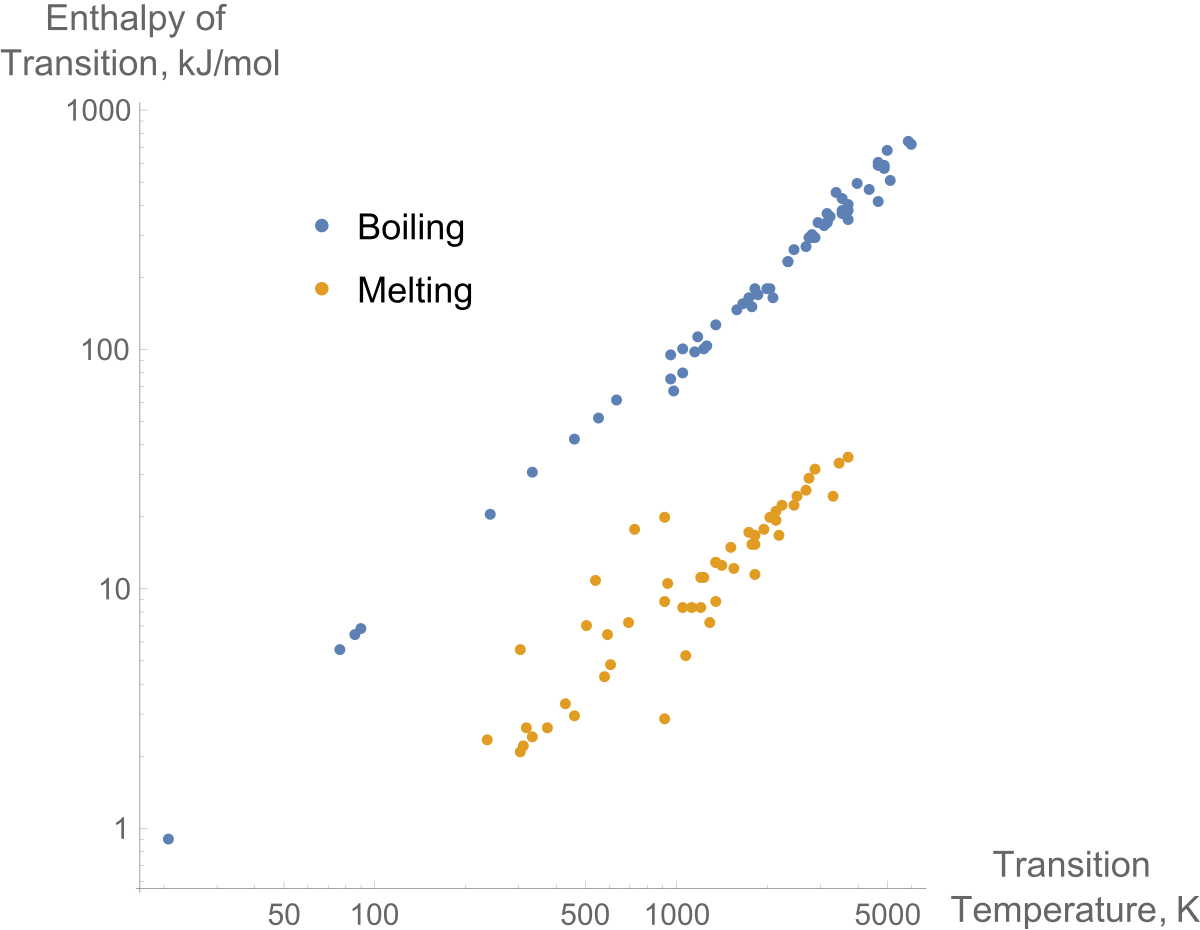
Enthalpy Of Water And Steam - Enthalpy of Wet Steam - Enthalpy of Dry And Saturated Steam - - YouTube

Calculate the enthalpy of formation of water, given that the bond energies of `H-H, O=O` and `O-... - YouTube

Calculate change in enthalpy when 2 moles of liquid water at 1 bar and `100^(@)` C is coverted into - YouTube

If water vapor is assumed to be a perfect gas, molar enthalpy change for vaporization of 1 mol of... - YouTube

Question Video: Calculating the Enthalpy of Dilution of Sodium Hydroxide Using Enthalpy of Solution Values | Nagwa