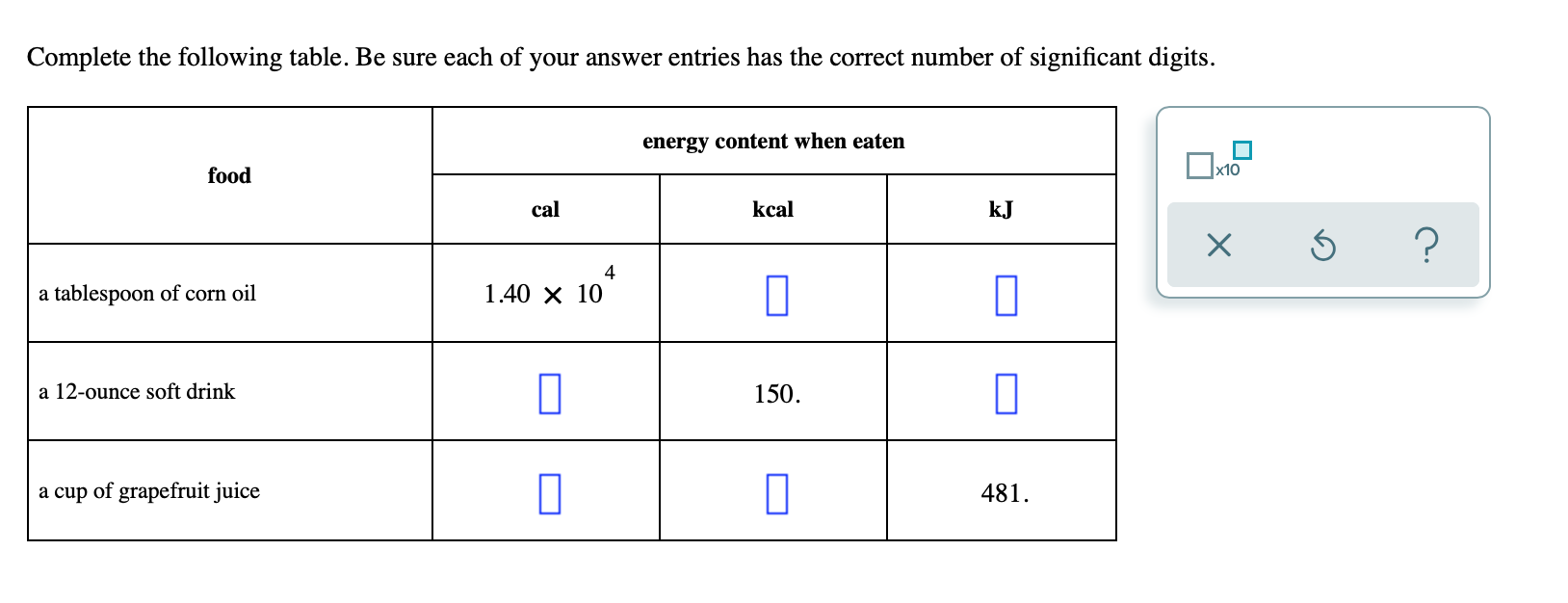
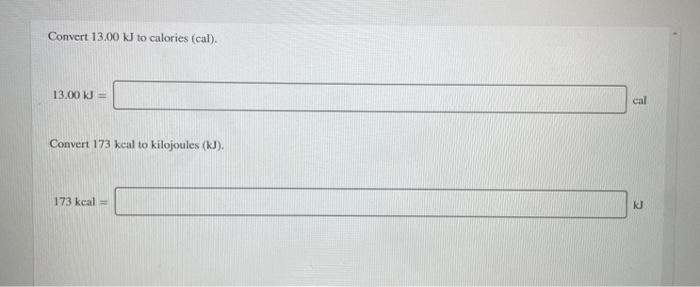
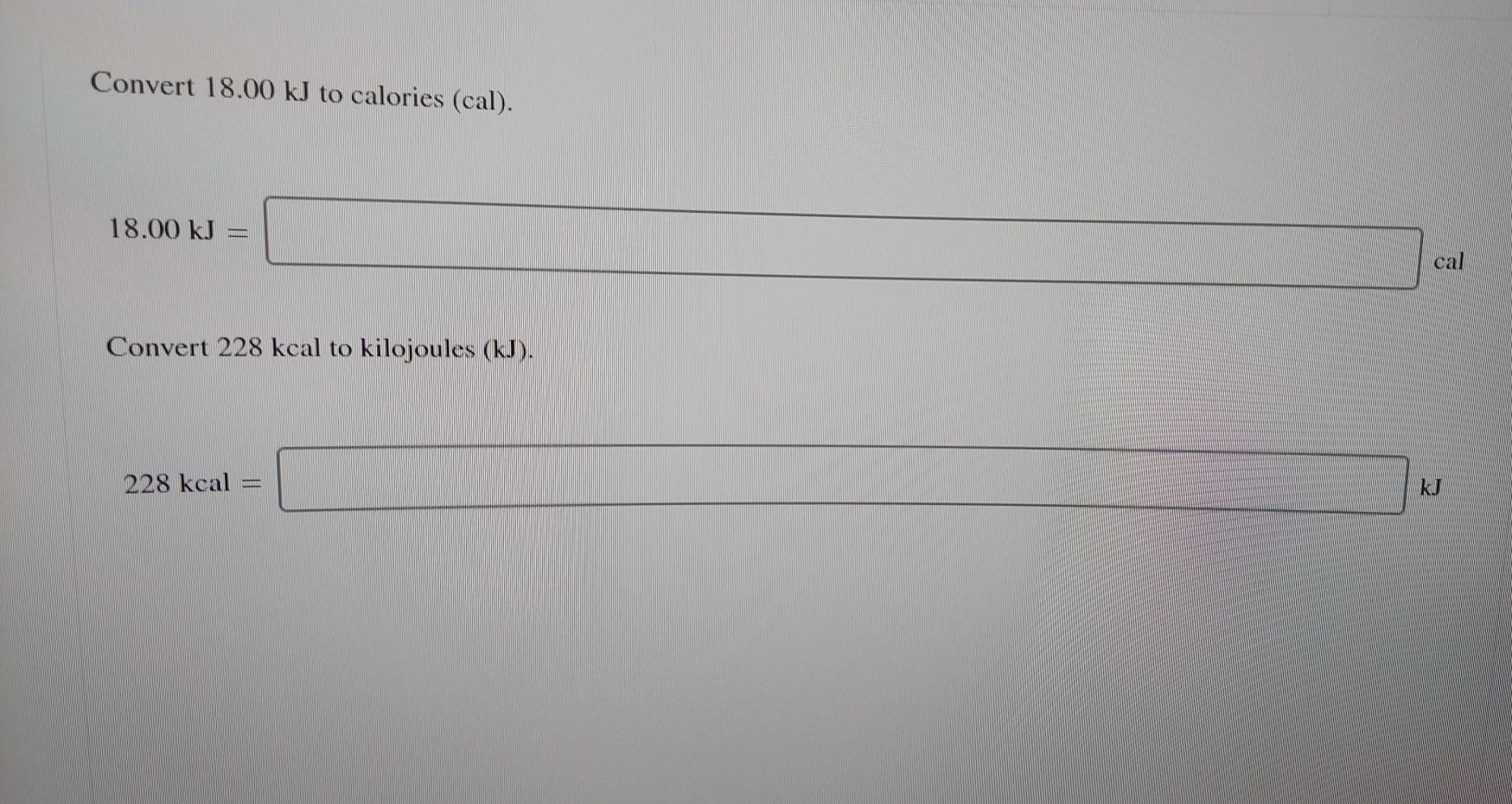
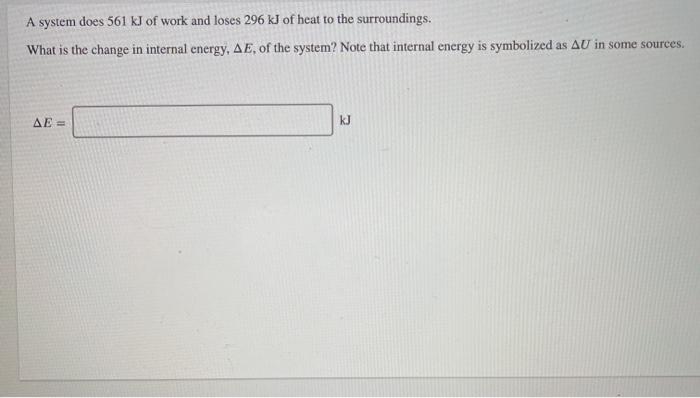
Given that C + O(2) rightarrow CO(2), Delta H^(@) = - x k J 2CO +O(2) rightarrow 2CO(2), Delta H^(@) = -y kJ What is heat of formation of CO?
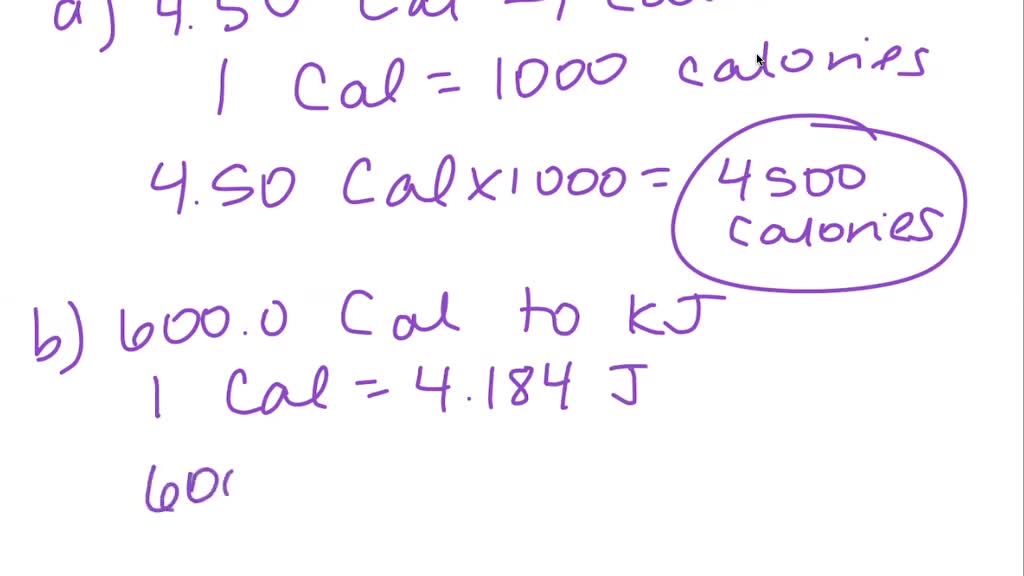
SOLVED:Convert: (a) 4.50 Cal to calories (b) 600.0 Cal to kilojoules (c) 1.000 J to calories (d) 50.0 Cal to joules
![TrainerRoad -> Strava Integration kJ vs. Calories [Feature Request] - TrainerRoad Software - TrainerRoad TrainerRoad -> Strava Integration kJ vs. Calories [Feature Request] - TrainerRoad Software - TrainerRoad](https://global.discourse-cdn.com/trainerroad/original/3X/f/6/f60d3c9bfdec3a63b1065d66230bd12c44f2dd9f.png)